Insulin Resistance: A Protocol for Prevention, Early Detection, and Reversal
Introduction
Insulin resistance is arguably the most significant health abnormality affecting humankind. Furthermore, the increasing prevalence of insulin resistance in adults, adolescents, and children highlights the growing threat that insulin resistance poses to individuals of all ages. While insulin resistance is typically associated with obesity, insulin resistance can also affect those with a normal body weight.1-3 To demonstrate this point, it is estimated that 16% of school-aged children with a normal body weight have insulin resistance.4 While it is widely recognized that insulin resistance contributes to type 2 diabetes, insulin resistance also contributes to a constellation of serious and disabling health abnormalities, including several types of cancer, dementia, liver disease, kidney disease, osteoarthritis, muscle loss, frailty, and infertility.5-12 Furthermore, there is evidence to suggest that insulin resistance is a stronger risk factor of cardiovascular disease than elevated levels of LDL-C and Apolipoprotein B (Link).13 Therefore, the early identification of insulin resistance is necessary for those seeking to promote longevity and avoid preventable medical illness.
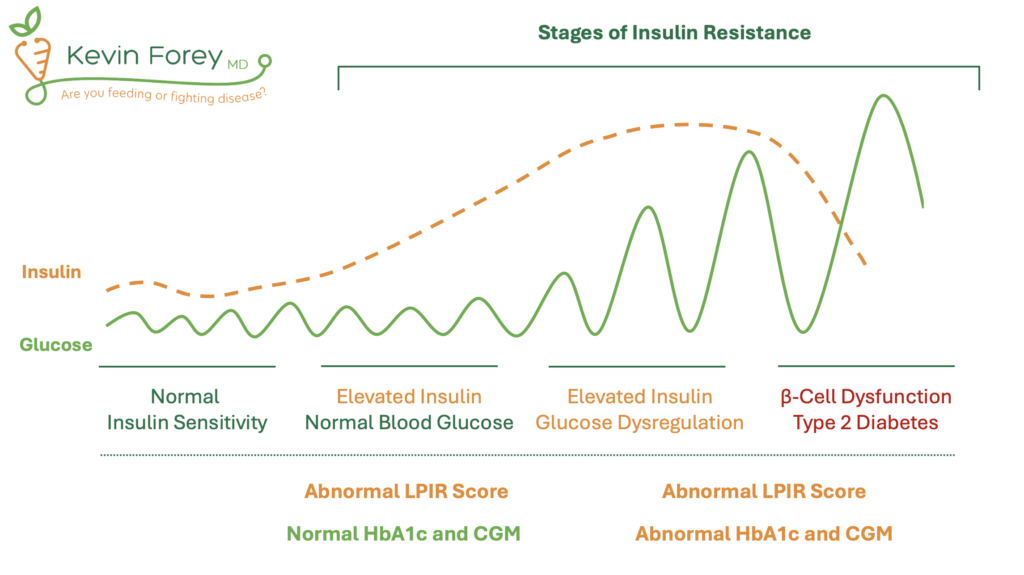
Meanwhile, Hemoglobin A1c (HbA1c) is the most commonly utilized test for assessing blood glucose control. However, HbA1c is not an optimal test for identifying insulin resistance. Specifically, it is a measurement of long-term blood glucose control and lacks the sensitivity to detect early stages of insulin resistance in those with normal blood glucose control (Figure 1).14,15 As a result, the standard approach to diagnosing insulin resistance using HbA1c often allows insulin resistance to go undiagnosed until significant health problems arise. Similarly, Continuous Glucose Monitors (CGMs) measure blood glucose fluctuations but may miss insulin resistance among those who are able to maintain normal blood glucose control, despite elevated levels of circulating insulin. Therefore, an optimal strategy is to utilize superior tests that identify insulin resistance prior to the onset of blood glucose dysregulation. This includes the LPIR Score, Triglyceride-Glucose Index (TyG Index), and HOMA-IR.
Importantly, insulin resistance is reversible in many cases, especially when addressed early with comprehensive lifestyle changes. Strategies to prevent insulin resistance are similar to the strategies used to reverse insulin resistance. This generally includes dietary modification, physical fitness, and the optimization of sleep and stress. In select instances, supplements and prescription medications can also be effective, however, optimizing food and nutrition and physical fitness is more effective than medications alone.16 Meanwhile, multiple strategies can be utilized simultaneously to maximize the likelihood of success. While the restriction of saturated fat is commonly recommended for the sake of reducing LDL cholesterol, this has no appreciable impact on improving insulin resistance.17 Therefore, we will explore evidence-based strategies capable of improving insulin resistance and other components of Metabolic Syndrome.
Related Podcast Episode
Disclaimer
This content is for general educational purposes only and does not represent medical advice or the practice of medicine. Furthermore, no patient relationship is formed. Please discuss with your personal healthcare professional before making any dietary, lifestyle, or medication changes. Additionally, I have no financial conflicts of interest or affiliations with any diagnostic testing or pharmaceutical companies mentioned in this article.
Notify Me of New Content
Provide your email address to receive notifications of new blog posts and podcast episodes.
Content Summary
What Is Insulin Resistance
Insulin resistance is an abnormal health condition in which the body fails to effectively respond to the hormone insulin. Under normal circumstances, the pancreas releases insulin into the bloodstream to regulate blood glucose levels. Insulin achieves blood glucose control by facilitating glucose uptake into liver, muscle, and fat tissue. It also plays a foundational role in directing fat storage and protein synthesis. The amount of insulin released by the pancreas is influenced by several factors, including the type of food consumed, the amount consumed, recent physical activity, stress levels, inflammation, pre-existing metabolic health, and other hormonal considerations.
In the case of insulin resistance, the body becomes less effective in its ability to respond to the hormone insulin. As a result, the body will compensate by releasing increased amounts of insulin into the circulation, a condition known as hyperinsulinemia. Importantly, hyperinsulinemia can occur before the onset of blood glucose dysregulation or elevations in blood glucose levels, implying that insulin resistance can exist before the onset of abnormalities seen in HbA1c or CGM.14,15 Meanwhile, even with normal blood glucose control, it is recognized that elevated levels of insulin contribute to a toxic health environment, resulting in chronic inflammation, organ damage, and cellular dysfunction. More specifically, while insulin resistance is widely recognized to contribute to type 2 diabetes, insulin resistance is a major risk factor of cardiovascular disease, several types of cancer, and dementia.5,6,31,32 Insulin resistance also contributes to liver disease, kidney disease, and the increasing need for dialysis and organ transplantation.7,8 Furthermore, insulin resistance is also associated with several disabling conditions, including osteoarthritis, muscle loss, frailty, and infertility (Table 2).9-12 Collectively, the United States spends more money treating the abnormalities of type 2 diabetes than any other health condition, including cardiovascular disease and cancer.33
Beyond insulin resistance is a broader health abnormality known as Metabolic Syndrome, which often manifests as some combination of excess abdominal obesity (visceral adiposity), high blood pressure (hypertension), elevated triglycerides, low HDL cholesterol, and inflammation. It is estimated that 85% of adults have at least one component of Metabolic Syndrome, with insulin resistance being the primary, underlying health abnormality of this broader condition.34 While many health experts and public health guidelines focus on body weight, often measured by Body Mass Index (BMI), and the restriction of calorie intake to improve body weight, Metabolic Syndrome is more strongly associated with cardiovascular disease than BMI alone (Table 2).13 Meanwhile, Metabolic Syndrome captures additional risk associated with poor metabolic health to a far greater degree than body weight or BMI alone. Therefore, it is advisable to view comprehensive metabolic health from the standpoint of Metabolic Syndrome, insulin resistance, and its relationship to abnormal lipoprotein (cholesterol) concentrations, rather than body weight and obesity alone. Once again, you do not have to be overweight to have insulin resistance or other abnormalities of Metabolic Syndrome.4
Table 1. Preventable Medical Illness Associated With Insulin Resistance
| Cancer | Colorectal Cancer | Breast Cancer | Pancreatic Cancer |
| Liver Cancer | Esophageal Cancer | Kidney Cancer | |
| Prostate Cancer | Endometrial Cancer | Ovarian Cancer | |
| Metabolic | Type 2 Diabetes | Liver Disease | Kidney Disease |
| Cardiovascular | High Blood Pressure | Heart Attack | Stroke |
| Autoimmune Disease | Ulcerative Colitis | Crohn’s Disease | Rheumatoid Arthritis |
| Inflammatory | Osteoarthritis | Gout | Acne |
| Neurological | Dementia | Parkinson’s Disease | Migraine |
| Reproductive | Erectile Dysfunction | Infertility + PCOS | Gestational Diabetes |
| Respiratory | Asthma | Sleep Apnea | Pulmonary Hypertension |
| Functional | Frailty | Muscle Loss | Osteopenia |
Table 2. Association of Risk Factors and Cardiovascular Disease13
| Risk Factor | Heart Disease Hazard Ratio, Age < 55 Years | Heart Disease Hazard Ratio, Age 65+ Years |
| Diabetes | 10.71 | 4.49 |
| Metabolic Syndrome | 6.09 | 2.82 |
| Hypertension | 4.58 | 2.06 |
| Obesity | 4.33 | 2.14 |
| Tobacco Use | 3.92 | 1.89 |
| Family History | 2.19 | 1.60 |
Early and Accurate Identification of Insulin Resistance
Because insulin resistance can exist before the onset of blood glucose dysregulation, traditional blood tests such as fasting glucose levels and HbA1c often fail to detect insulin resistance in its early stages.14,15 As a result, insulin resistance may exist for years or decades before abnormalities in HbA1c, fasting blood glucose, or CGMs are observed.
Furthermore, HbA1c can also be elevated in healthy individuals without insulin resistance. Perhaps the most common circumstance in which a healthy individual may have an elevated HbA1c includes endurance athletes who are observed to have higher fasting glucose levels, likely due to the body’s regular demand for circulating blood glucose during times of prolonged physical activity, resulting in upregulated gluconeogenesis (increased production of glucose in the liver and kidneys).35 Other potential factors include those with a prolonged red blood cell lifespan, iron deficiency, and certain medications.
These examples of the limitations of HbA1c underscore the importance of utilizing tests that more directly assess insulin resistance compared to Hemoglobin A1c. Rather than utilizing a single biomarker alone, a thorough and comprehensive evaluation of insulin resistance should utilize the combination of the LPIR Score, TyG Index, HOMA-IR, and other assessments of metabolic health (Table 3).
- Lipoprotein Insulin Resistance Score (LPIR Score): A useful blood test that analyzes changes in lipoprotein particle size and concentration to determine the presence and severity of insulin resistance. It is particularly helpful in detecting early stages of insulin resistance, including those with a normal body weight and normal blood glucose control.18,19 Notably, the LPIR Score was developed and validated using data from the euglycemic hyperinsulinemic clamp, which is the most precise method for measuring insulin resistance (“the gold standard”). Because the euglycemic hyperinsulinemic clamp is labor-intensive, it is only used in research settings and not available as a routine laboratory test. Instead, the LPIR Score bridges this gap by providing a simple blood test that is a highly reliable surrogate marker of insulin resistance, and calibrated to the gold standard for measuring insulin resistance.
- Triglyceride-Glucose Index (TyG Index): This index is calculated using a formula that combines fasting triglyceride and fasting glucose levels, making it both cost-effective, accessible, and another useful test for detecting insulin resistance.20 Research has demonstrated the TyG Index to be a superior predictor of insulin resistance compared to HbA1c and HOMA-IR.20 Beyond insulin resistance, the TyG Index is strongly associated with lifespan (all-cause mortality), frailty, multiple types of cancer including breast and colon cancer, and dementia.21-27
- HOMA-IR: Another widely used test that utilizes fasting glucose and fasting insulin levels. While HOMA-IR is highly sensitive, it is also susceptible to large day-to-day fluctuations in fasting insulin and glucose levels that may be influenced by factors unrelated to insulin resistance.28-30 Therefore, HOMA-IR should be interpreted in conjunction with other testing strategies to provide a comprehensive analysis of insulin resistance.
- Hemoglobin A1c (HbA1c): A measurement of long-term blood glucose control, not insulin resistance. While there is a strong degree of correlation between blood glucose dysregulation and insulin resistance, the use of HbA1c is best for monitoring long-term blood glucose control in those with advanced states of insulin resistance and type 2 diabetes. Meanwhile, it appears less helpful for detecting early stages of insulin resistance.
Table 3. Early and Accurate Identification of Insulin Resistance
| No Insulin Resistance | Elevated Glucose Possibly Unrelated To Insulin Resistance | Early Insulin Resistance | Early to Advanced Insulin Resistance +/- Diabetes |
| Normal HbA1c | Slightly Elevated HbA1c | Normal HbA1c | Abnormal HbA1c |
| Normal LPIR Score, TyG Index, and HOMA-IR | Normal LPIR Score, TyG Index, and HOMA-IR | Abnormal LPIR Score, TyG Index, and/or HOMA-IR | Abnormal LPIR Score, TyG Index, and/or HOMA-IR |
Figure 1. Stages of Insulin Resistance
Strategies to Prevent and Reverse Insulin Resistance
There are a variety of effective strategies for the prevention and treatment of insulin resistance. This includes dietary modification, physical fitness, optimization of sleep, stress management, and in some instances, supplements and prescription medications. The same strategies used to reverse insulin resistance are typically effective for the prevention of insulin resistance. However, among those who are metabolically healthy and without evidence of insulin resistance, there is more leniency in the degree of adherence to these strategies necessary to prevent insulin resistance. Meanwhile, for those with insulin resistance, poor metabolic health, and/or a family history of insulin resistance, it is reasonable to implement as many of these strategies as possible. Additionally, it is advantageous to remain as disciplined as possible in following these strategies. Meanwhile, all individuals will respond differently to these interventions, and professional guidance from a licensed healthcare professional is necessary for ensuring safety.
Food, Nutrition, and Physical Fitness
When evaluating the relative effectiveness of aerobic exercise, strength training, and dietary modification, all are effective strategies for reversing and preventing insulin resistance. However, clinical trials have demonstrated that dietary modification is the single most impactful strategy for reversing insulin resistance (Figure 2).38 Meanwhile, moderate-intensity aerobic exercise appears to be equally as effective, or more effective, than vigorous-intensity aerobic exercise. Both moderate-intensity and vigorous-intensity aerobic exercise appear more effective than strength training in isolation. Interestingly, the combination of aerobic exercise and strength training demonstrates a positive synergistic effect, for which the combined benefit of aerobic exercise and strength training appear greater than the individual benefits of each modality. Importantly, the greatest benefits in overcoming insulin resistance are achieved with the combination of maximizing and optimizing food and nutrition, aerobic exercise, and strength training.
Figure 2. Impact of Aerobic Exercise, Strength Training, and Dietary Modification on Insulin Sensitivity38
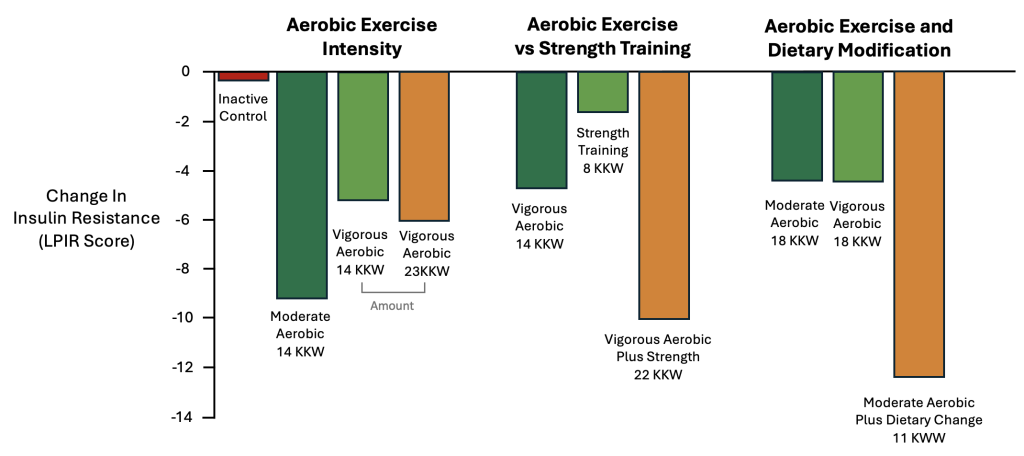
Note: The unit kcal/kg of body weight/week (KKW) is a measure of the prescribed amount of exercise in terms of energy expenditure relative to body weight over a one-week period.
Optimizing Food and Nutrition
Food and nutrition likely represents the most important modifiable risk factor regarding insulin resistance and the array of health abnormalities associated with Metabolic Syndrome. Meanwhile, the subject of food and nutrition is highly controversial, with conflicting perspectives and nutritional priorities. While some health experts take a cautious approach, offering broad, non-controversial recommendations, such as maintaining a healthy energy balance, follow a Mediterranean diet, and prioritize physical activity, a more precise and scientific approach is required to fully address the complexities of metabolic health and insulin resistance.
The Toxic Food Hypothesis
Over the past century, rates of obesity, insulin resistance, and Metabolic Syndrome have risen dramatically, largely due to fundamental changes in the food system.39,40 The transition from naturally occurring whole foods to a diet dominated by highly processed products now accounts for nearly 60% of total calorie intake in the United States.41 Despite growing evidence linking highly processed foods to poor metabolic health, mainstream nutrition guidelines continue to emphasize a “balanced diet” without adequately addressing the unique harms of these foods. The common recommendation to “eat everything in moderation” fails to acknowledge that some foods are inherently detrimental to human health. Rather than moderation, a more effective approach is to minimize or eliminate the regular consumption of highly processed foods.
The Toxic Food Hypothesis proposes that highly processed foods disproportionately contribute to insulin resistance through two primary mechanisms:
- Overconsumption: Processed foods promote excessive calorie intake compared to whole foods.42
- Metabolic Dysfunction: Even at equal caloric intake, highly processed foods uniquely damage metabolic health.43
The Toxic Food Hypothesis challenges the conventional calorie-centric model, emphasizing that food quality, not just quantity, is critical for metabolic health. Addressing insulin resistance requires more than simply adding healthier foods, it necessitates a fundamental shift away from highly processed foods.
Defining Highly Processed Foods
A common critique of the Toxic Food Hypothesis is the difficulty of defining “highly processed foods.” While classification systems like NOVA’s “ultra-processed foods” exist, there are several notable limitations and shortcomings. Given the vast diversity and complexity of the modern food supply, rigid definitions are impractical. A typical grocery store carries more than 30,000 food products, for example. Instead, a more useful approach is to consider processed foods qualitatively along a spectrum:
Naturally Occurring → Minimally Processed → Highly Processed → Ultra-Processed
For this discussion, highly processed foods refer to products that cannot be made at home using simple ingredients and basic food preparation techniques. Importantly, cooking is not the same as industrial food processing. Unlike household food preparation techniques (e.g., chopping vegetables, heating food, grinding peanuts to make peanut butter), industrial food processing involves chemical modifications and additives that fundamentally alter food composition. While not all processed foods are harmful, many contribute to poor metabolic health.
Strategies for Optimizing Food and Nutrition:
- Minimize or Eliminate Added and Refined Sugars: A deliberate avoidance of highly processed foods, including highly processed sugars and carbohydrates, likely represents the greatest health opportunity for the majority of individuals with insulin resistance. To optimize health, individuals should aim to reduce or avoid added sugars found in sweets, candy, sodas, energy drinks, flavored coffee drinks, and sugar-containing condiments. Sugars contained in fruit juices should also be avoided, even if “all natural” or “organic.” Additionally, highly refined carbohydrates containing sugar should be avoided, including pastries, muffins, breakfast cereals, and many processed snack-foods. Among those with insulin resistance, restricting added and refined sugar to less than 15 grams per day is a reasonable goal.
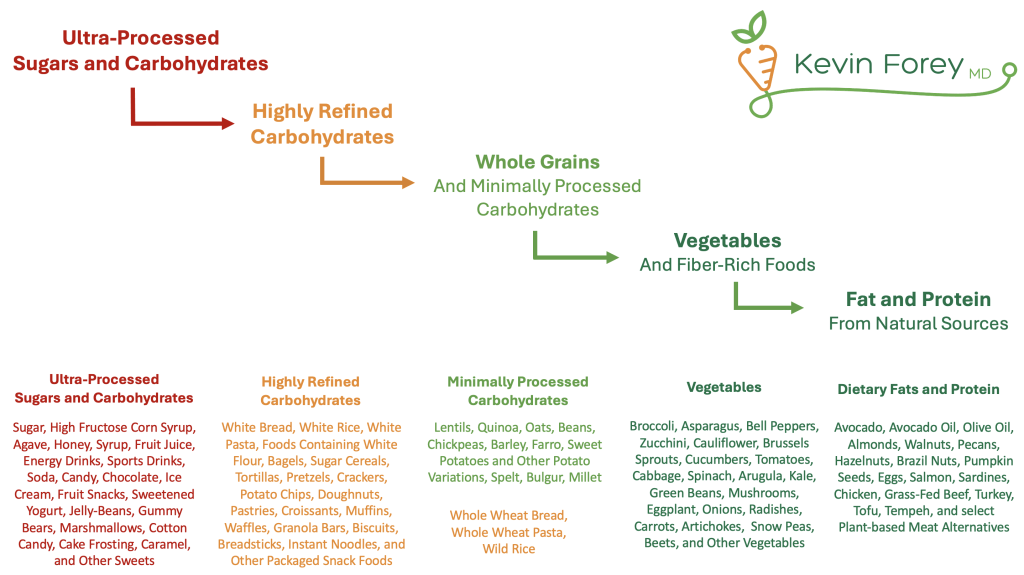
- Improve Carbohydrate Quality: Significant health improvements can be made by focusing on the quality of the calories we consume. Research has shown that simply replacing high-fructose corn syrup with an equal amount of calories from starch-based carbohydrates can lead to noticeable improvements in insulin resistance, blood pressure, body weight, inflammation, and LDL cholesterol (Figure 3).43 These benefits were observed within just two weeks, without any reduction in total calorie or carbohydrate intake. Instead, the key change was a deliberate shift toward higher-quality carbohydrates and the elimination of added and refined sugars.
Practical Examples to Improve Carbohydrate Quality:
- Replace soda → flavored sparkling water.
- Replace pasta, sandwiches, and grains → salad; eat more salad!
- Replace french fries and onion rings → sautéed or grilled vegetables.
- Replace white rice → riced cauliflower, whole grains, or legumes.
- Replace chips → mixed nuts, carrots and almond butter, or sliced peppers and hummus.
- Replace granola → mixed nuts with cinnamon.
- Replace a sugar-containing dessert → mixed fruit.
Note: Be mindful of the sugar content in common fruits; those with insulin resistance should preferentially select fruits with low sugar content (Table 5).44
Figure 3. Stepwise Approach to Improving Carbohydrate Quality
- Advanced Analysis of Carbohydrate Quality: To better understand carbohydrate quality, several advanced nutritional metrics can offer valuable insight. Key among these are the Insulin Index and Glycemic Load (not the Glycemic Index), both of which are particularly useful in assessing food choices for individuals with insulin resistance (Table 4). Choosing foods that are low in both Glycemic Load and Insulin Index can lead to significant improvements in insulin sensitivity. 45-48 In addition, other useful metrics include the Percent Fiber of Total Carbohydrate and the Protein-to-Carbohydrate Ratio (Table 4). While daily monitoring of these metrics is not necessary, they serve as useful reference points to scientifically evaluate food choices, illustrating a clear pattern from naturally occurring to highly processed foods, as shown in Table 4.
While the Glycemic Index is widely understood and commonly used, it is less reliable for daily dietary decisions. Research suggests that alternative methods like Glycemic Load offer more consistent outcomes regarding the improvement of insulin resistance.49
Table 4. Advanced Analysis of Carbohydrate Quality per 100 grams (Abbreviated Version)
| Food Item | Net Carbohydrates (g) | % Fiber of Total Carbs | Carb:Protein Ratio | Glycemic Load (GL) | Insulin Index | Energy Density (kcal/100g) |
| Jelly Beans | 90 | 0% | No Protein | 80 | 160 | 380 |
| Pretzels | 80 | 3% | 7:1 | 60 | 100 | 380 |
| White Bread | 44 | 3% | 5:1 | 45 | 100 | 265 |
| Whole Wheat Bread | 40 | 7% | 4:1 | 35 | 80 | 252 |
| Quinoa | 18 | 13% | 4:1 | 12 | 50 | 120 |
| Lentils | 12 | 40% | 1.3:1 | 8 | 40 | 116 |
| Broccoli | 4 | 37% | 1.3:1 | 1 | 18 | 34 |
| Spinach | 1 | 60% | 1:3 | < 1 | 12 | 23 |
Note: Definitions of the above terms and measurements are discussed in Table 11. Values are approximate estimates from a variety of online sources including Food Struct.
Table 5. Sugar content of common fruits.
| High Sugar (> 12g per 100g) | Moderate Sugar (7-12g per 100g) | Low Sugar (< 7g per 100g) |
| Dates – 66.5g | Apples – 10.4g | Watermelon – 6.2g |
| Figs (dried) – 48.6g | Pineapple – 9.9g | Cantaloupe – 5.7g |
| Grapes – 16.3g | Pears – 9.8g | Strawberries – 4.9g |
| Mango – 14.8g | Blueberries – 9.7g | Blackberries – 4.9g |
| Cherries – 13.0g | Oranges – 9.2g | Raspberries – 4.4g |
| Bananas – 12.2g | Kiwi – 8.9g | Avocado – 0.7g |
Note: This article is not meant to demonize fruit. However, clinical research has demonstrated that increasing fruit intake, without making other dietary changes, can worsen insulin resistance, blood sugar control, and liver inflammation, in individuals with poor metabolic health at baseline.44
- Reduce Total Carbohydrate Intake: In addition to improving the quality of carbohydrates consumed, reducing total carbohydrate intake is often necessary for individuals with advanced insulin resistance, metabolic syndrome, fatty liver disease, and type 2 diabetes. Well-designed clinical trials have shown that carbohydrate restriction is an effective strategy for improving insulin resistance.50-54 Because highly processed foods are disproportionately composed of highly refined carbohydrates, most studies evaluating “Carbohydrate Restriction” or “Low-Carbohydrate Diets” are indirectly evaluating the impact of restricting highly processed foods.
Although reducing saturated fat is commonly recommended for lowering ApoB and LDL-C levels, it does not meaningfully improve insulin resistance.17 In randomized clinical trials, Carbohydrate Restriction and the Mediterranean Diet often outperform Low-Fat Diets.50,55 When compared directly, Carbohydrate Restriction may be equal to or even more effective than the Mediterranean Diet.50,56-58 Importantly, the Mediterranean Diet shares several key components with many forms of Carbohydrate Restricted Diets, including (1) increased intake of dietary fats from natural sources like mixed nuts and olive oil, (2) reduced consumption of highly refined carbohydrates, and (3) greater focus on whole grains and legumes.
How closely an individual adheres to carbohydrate restriction depends on their unique circumstances and healthcare goals. For those with insulin resistance, it is important to discuss with a licensed healthcare professional to determine safe and appropriate targets. A common strategy is to reduce total carbohydrate intake by about 20-30%. For context, the average American adult consumes at least 240 grams of carbohydrates per day, with adult males averaging 260 to 300 grams per day.59-60 Additionally, the intake of added and refined sugars should be limited to less than 10 to 15 grams per day. Research by Christopher Gardner, Ph.D., and colleagues has shown that significant improvements in insulin resistance can be achieved with a total carbohydrate intake of less than 140 grams per day, with additional benefits from further restriction (Table 6).61 However, the quality of carbohydrates remains the most important factor, with a focus on minimizing or eliminating added sugars, refined sugars, and highly processed carbohydrate-rich foods.
This article is not intended to advocate for a single approach, but rather, to emphasize the importance of avoiding highly processed foods, especially added and refined carbohydrates, which disproportionately contribute to insulin resistance.
Table 6. Impact of Carbohydrate Intake on Biomarkers of Insulin Resistance in the A to Z Trial61
| Energy From Carbohydrate, % (grams per day) | 17.7 % (61 g/day) | 29.5 % (113 g/day) | 34.5 % (138 g/day) |
| Insulin, µU/mL | – 3.0 µU/mL | – 2.8 µU/mL | – 1.8 µU/mL |
| Triglycerides, mg/dL | – 52.3 mg/dL | – 35.6 mg/dL | – 29.3 mg/dL |
| Triglyceride-Glucose Index | – 0.27 | – 0.17 | – 0.15 |
| HOMA-IR | – 1.6 | – 1.6 | – 0.5 |
- Consider Time-Restricted Eating: Both calorie restriction and time-restricted eating can improve insulin resistance, but emerging evidence suggests that time-restricted eating may offer distinct advantages. Some studies have demonstrated that time-restricted eating alone can result in an improvement in insulin sensitivity, pancreatic beta-cell function, and metabolic health, even without a reduction in calorie intake.62 Other clinical trials have achieved improvements in metabolic health utilizing time restricted eating, particularly when meals are consumed earlier in the day, aligning with circadian rhythms.63 Importantly, the evidence remains mixed and more research is needed to confirm whether or not there are unique benefits of time-restricted eating over calorie restriction.64,65
In theory, at a cellular level, a prolonged duration of fasting will promote greater improvements and recovery of insulin resistance when no calories are being consumed and no insulin is being secreted. In contrast, caloric restriction continues to stimulate insulin secretion, even when calorie intake is reduced. Therefore, as we await additional scientific research, it remains plausible that time-restricted eating may offer unique benefits for the sake of improving insulin resistance. Meanwhile, caloric restriction and time-restricted eating are both effective strategies for improving insulin resistance.
- Track Glucose Levels Continuously: Continuous Glucose Monitors (CGMs) provide real-time feedback regarding glucose fluctuations in response to food intake, physical activity, stress, sleep, and medications. By offering instant, visual feedback, CGMs provide personalized data that can help to influence positive lifestyle changes. For example, if a particular food causes a glucose spike, choosing an alternative food or reducing the portion size may help to improve insulin resistance. This is particularly helpful and relevant to those with insulin resistance, where real-time, day-to-day feedback of food choices and the resultant blood glucose control can help to inform and modify daily decision making.
Meanwhile, in individuals without insulin resistance, the role of CGMs remains less clear due to the lack of reproducibility and consistency in CGM reported blood glucose values.66 Furthermore, glucose spikes to 140 to 150 mg/dL are normal and should not necessarily cause alarm and anxiety. Recent scientific research has acknowledged the reality that CGMs can overestimate the prandial glucose response when compared to capillary finger sampling.67 Given that lack of clear thresholds, issues with reproducibility, and issues with accuracy, CGMs should not be used to diagnose insulin resistance, but should instead provide insight into those with blood glucose dysregulation and the need to further evaluate the possibility of insulin resistance.
Balancing ApoB, Insulin Resistance, and Individual Circumstances
Reductions in carbohydrate intake often leads to an increased consumption of dietary fats, including saturated fats. While the degree of carbohydrate processing significantly influences health, the same applies to dietary fats, where the source and composition matter. Importantly, higher saturated fat intake can elevate ApoB levels,68 however, this effect varies considerably69 depending on the type and quality of saturated fat consumed,70 as well as individual biological and genetic factors. Given these complexities, individuals making dietary changes should monitor blood work, including ApoB, to assess their individual response. For those looking to optimize ApoB levels or mitigate potential increases associated with carbohydrate restriction, several dietary strategies can be considered:
- Increase Fiber Intake: Increasing fiber intake, particularly soluble fiber, is an effective strategy for lowering LDL-C and ApoB. Soluble fiber found in foods such as psyllium, oats, barley, beans, and lentils, binds to bile acids and cholesterol, reducing absorption and increasing excretion, which in turn leads to greater LDL-C clearance and reduced ApoB levels. Clinical studies suggest that consuming 10 grams of soluble fiber per day can lower LDL-C by approximately 10 mg/dL.71,72 Insoluble fiber, found in wheat bran, whole grains, nuts, and vegetables, has also been shown to lower LDL-C and ApoB by increasing fecal bulk and reducing intestinal transit time. While its effects on ApoB are less pronounced than those of soluble fiber, it is another strategy for lowering ApoB.73,74
- Limit Consumption of Saturated Fat From Highly Processed Foods: Saturated fats from highly processed foods can raise ApoB levels. These include processed meats, processed cheese products (e.g., American cheese, cheese spreads), many ready-to-eat and frozen meals, packaged snacks, certain baked goods, and fast food restaurants. Reducing intake of industrially manufactured and processed foods is a reasonable strategy for optimizing saturated fat intake and ApoB levels.
- Replace Butter and Coconut Oil with Avocado Oil: When cooking, replacing butter or coconut oil with avocado oil reduces saturated fat intake while avoiding reliance on highly processed oils like canola or soybean oil. Avocado oil is predominantly composed of monounsaturated fats, which are associated with favorable lipid profiles and remains stable at high cooking temperatures.
- Limit Consumption of Industrial Raised Livestock: Industrial-raised, grain-fed livestock have a less favorable fatty acid profile than grass-fed livestock. Grass-fed beef and dairy, for example, contain 2–5 times higher concentrations of omega-3 fatty acids, a higher proportion of stearic acid (which has a neutral effect on LDL-C and ApoB), and greater levels of antioxidants compared to grain-fed livestock.75,76 Preferentially selecting high-quality, naturally raised animal products is another approach to improving the quality and composition of dietary fat consumed.
- Consider Select Plant-Based Meat Alternative Products: Some plant-based meat alternatives, such as tofu and tempeh, are minimally processed and provide beneficial metabolic effects. However, newer plant-based substitutes vary widely in ingredient quality and processing methods. While some use whole-food ingredients and natural seasonings, others contain industrially processed components similar to traditional processed foods. Some studies suggest that plant-based meat alternatives may contribute to favorable metabolic health outcomes, including improvements in insulin resistance and reductions in LDL-C.77,78 However, not all studies have demonstrated improvements in LDL-C,79 and the impact on ApoB remains less well studied. When incorporating these products, selecting options with minimal additives and a whole-food-based ingredient list is preferable.
Aerobic Exercise and Strength Training
Among numerous other benefits, physical fitness is one of the most effective interventions for preventing and reversing insulin resistance. Both aerobic and resistance training offer unique but complementary benefits, making a combined approach ideal for optimizing metabolic health and insulin sensitivity.
Aerobic Exercise
Aerobic exercise enhances insulin sensitivity through multiple mechanisms, including the upregulation of GLUT4 transporters, which facilitate insulin-independent glucose uptake, which is particularly helpful for individuals with impaired insulin sensitivity. In other words, aerobic exercise stimulates alternative pathways for glucose management, partially bypassing the limitations imposed by existing insulin resistance. Additionally, aerobic training enhances mitochondrial biogenesis and efficiency, which can improve insulin signaling. Aerobic exercise also increases glycogen storage capacity and improves skeletal muscle glucose disposal. Among other benefits, aerobic exercise also stimulates nitric oxide production, which enhances vascular function, muscle perfusion, and glucose delivery. Collectively, these physiological adaptations make aerobic exercise a powerful intervention for preventing and reversing insulin resistance.80-82
Practical Strategies to Optimize Aerobic Exercise:
- Prioritize Total Training Time: While there is tremendous interest and enthusiasm regarding heart rate zones and aerobic exercise, research has demonstrated that the total duration of aerobic exercise is a more significant factor in improving cardiorespiratory fitness and metabolic health.83 Although individual targets and training goals will vary, current guidelines recommend at least 150 minutes per week of moderate-to-vigorous exercise. Notably, longer durations of aerobic exercise continue to provide benefits, with no clear upper limit for most individuals.83,84
- Maximize Moderate-Intensity Training: Research suggests that moderate-intensity aerobic exercise (Zone 2) may be as effective, or even more effective, than high-intensity exercise in improving insulin resistance.38 While any intensity of aerobic exercise benefits metabolic health, training at 60 to 70% of your maximum heart rate (Zone 2) has been shown to enhance mitochondrial function and metabolic flexibility. Although precise definitions and thresholds for Zone 2 exist, it can generally be approximated as the intensity at which you can maintain a conversation, though it feels somewhat strained or out of breath.
- Incorporate High-Intensity Interval Training: Incorporating short bursts of high-intensity aerobic exercise (85–95% of max heart rate) with recovery periods offers a time-efficient strategy to enhance glucose uptake and insulin sensitivity. Combining both moderate- and high-intensity aerobic exercise optimizes their distinct benefits, including improved cardiorespiratory fitness (VO2 max), enhanced mitochondrial function, better endothelial health, and positive cardiovascular adaptations.
- Avoid Stagnation and Digression: Long-term improvements require progressive adaptation. Gradually increasing duration, frequency, or intensity prevents metabolic stagnation and ensures sustained improvements in insulin sensitivity and metabolic health. Remaining at the same effort level indefinitely can result in diminishing returns.
Strength Training
Strength training enhances insulin sensitivity through several mechanisms, primarily by increasing muscle mass, which serves as the largest site for glucose uptake and storage. Since skeletal muscle is the primary tissue for insulin-mediated glucose disposal, greater muscle mass creates a larger reservoir for glucose utilization. Additionally, strength training increases GLUT4 expression, improving the efficiency of insulin-dependent glucose uptake. It has also been shown to enhance capillary density, which improves muscle perfusion and glucose delivery. These adaptations make strength training an effective strategy for both preventing and managing insulin resistance.
Practical Strategies to Optimize Strength Training:
- Maximize Training Volume: The total training volume (weight × repetitions × sets) is the single most important determinant of strength training benefits. In other words, lifting light weights with higher repetitions can achieve comparable benefits as lifting heavier weights with fewer repetitions.85 For beginners, starting with lower weights and higher repetitions is a safe and effective approach that may reduce risk of injury.
- Prioritize Large Muscle Groups: To optimize metabolic health and improve insulin sensitivity, strength training should target the largest muscle groups in the body. Exercises that engage the legs, back, and chest have the most significant positive impact on metabolic health. For individuals with limited time or less frequent strength training sessions, prioritizing leg and back exercises can maximize benefits for insulin sensitivity. However, incorporating full-body strength training remains the ideal approach for overall metabolic health and functional strength.
- Prioritizing Multi-Joint Exercises: Complex, multi-joint exercises provide superior benefits compared to machine-based strength training.85 Movements like squats, deadlifts, bench presses, push-ups, and pull-ups, engage multiple muscle groups simultaneously, which enhance movement patterns, neuromuscular coordination, and stability more effectively than isolated machine-based exercises. While machine-based exercises can be useful for targeting specific muscles, multi-joint movements provide more benefit in less time.
- Practice Progressive Overload: Gradually increasing resistance, whether by adding weight, repetitions, or sets, ensures continued adaptation and helps prevent metabolic plateaus. If completing 10–15 repetitions feels too easy, the weight is likely insufficient, and a heavier load should be considered. Additionally, progressively increasing the weight with each successive set (e.g., adding 5 or 10 lbs) can further enhance strength gains and metabolic benefits.
- Focus on Eccentric and Concentric Movements: Strength training consists of two key phases: muscle contraction (concentric) and muscle elongation (eccentric). Performing both phases in a slow, controlled manner enhances muscle activation and training effectiveness. To maximize strength training, avoid rapid movements, resist gravity, and maintain control throughout each repetition.
- Maintain Proper Form and Minimize Injury Risk: Proper biomechanics are essential for injury prevention and long-term success. Focus on controlled movements, proper alignment, and sufficient recovery to sustain progress. Avoidable injuries can have lasting consequences, potentially accelerating physical decline and contributing to premature frailty.
Hybrid Exercises for Optimizing Metabolic Health
Combining aerobic and resistance training provides synergistic benefits, enhancing insulin sensitivity, aerobic capacity, and overall strength. Integrating exercises that engage both cardiovascular and muscular systems maximizes efficiency, metabolic adaptation, and functional performance.
Hybrid Exercises for Improving Insulin Sensitivity
- Stair Climber: A weight-bearing exercise that strengthens the gluteal muscles, quadriceps, and calves, while simultaneously improving cardiovascular endurance.
- Sled Push: A high-intensity, full-body exercise that enhances lower-body strength, power, and cardiovascular endurance.
- Farmer’s Carry: A functional strength exercise that improves grip endurance, core stability, and overall metabolic conditioning.
- Rowing Machine: A low-impact, high-efficiency exercise that combines cardiovascular conditioning with full-body strength development, targeting the legs, core, and upper body.
- Kettlebell Swings: A dynamic full-body movement that enhances cardiovascular endurance while strengthening the glutes, hamstrings, and lower back.
- Battle Ropes: A high-intensity, full-body workout that enhances cardiovascular fitness while building upper-body strength and muscular endurance.
Optimizing Sleep and Stress
Sleep impairment and uncontrolled psychological stress both contribute to insulin resistance through a combination of hormonal imbalances, inflammation, and metabolic dysfunction. Furthermore, there is a notable relationship between sleep, stress, and psychological health, where sleep impairment can aggravate psychological health and vice versa. Among these factors, sleep impairment has emerged as a major contributing risk factor of insulin resistance, with research consistently demonstrating that chronic sleep deprivation, impaired sleep quality, and abnormal sleep patterns, all contribute to insulin resistance and adverse metabolic outcomes.86 This article will briefly evaluate the evidence regarding insulin resistance and its relationship to sleep quantity, sleep quality, and circadian regularity.
Sleep Quantity
In a randomized crossover study, a 1.5-hour nightly sleep reduction resulted in impaired insulin sensitivity measured by increased fasting insulin levels and higher HOMA-IR values.87 Notably, these changes occurred without alterations in body fat, suggesting that sleep itself plays a direct role in insulin signaling and glucose regulation. In a separate study, four days of sleep restriction resulted in impaired insulin signaling, with a 19% reduction in insulin sensitivity measured via intravenous glucose tolerance test compared to normal sleep sleep conditions.88 Similar findings were reported in a separate randomized crossover study, where five nights of sleep restriction reduced whole-body insulin sensitivity by 25%, peripheral insulin sensitivity by 29%, increased cortisol by 21% (stress hormone), and circulating fatty acids by 62%.89 Importantly, even a single night of sleep restriction of 4-hours has been demonstrated to reduce insulin sensitivity by approximately 25% in healthy adult subjects.90 Collectively, these findings emphasize that inadequate sleep quantity, even in the short term, can have profound effects on insulin sensitivity.
Sleep Fragmentation (Quality)
Beyond sleep duration, poor sleep quality can increase the risk of insulin resistance, with common examples including frequent awakenings, sleep disruptions, and reduced deep sleep. Sleep fragmentation occurs when sleep is interrupted by frequent arousals, even if total sleep time remains adequate. Clinical research has convincingly demonstrated that fragmented sleep impairs insulin sensitivity, glucose metabolism, and normal sympathetic nervous system activity. This heightened stress response reduces insulin’s effectiveness, pushing the body toward insulin resistance. Common examples of sleep fragmentation include noise disruption, light disruption, obstructive sleep apnea, restless leg syndrome, chronic insomnia, or other sleep disorders.
To demonstrate the negative health effects of sleep fragmentation, healthy adults were exposed to disruptive auditory stimuli for 3 nights, with a 25% reduction in insulin sensitivity observed via euglycemic clamp, and corresponding elevations cortisol and norepinephrine levels.91 In a separate example among individuals with obstructive sleep apnea, three months of CPAP therapy significantly improved insulin sensitivity measured by HOMA-IR.92 A mechanistic explanation was that CPAP therapy reduced nocturnal hypoxia (low blood oxygen) and sleep fragmentation, for which intermittent hypoxia and night-time arousals both contribute to increased sympathetic activity and inflammation (e.g., IL-6) with resultant damage to insulin sensitivity.
Circadian Regularity
In addition to sleep duration and quality, the timing and regularity of sleep are important. Specifically, clinical research has demonstrated that late bedtimes, described as circadian misalignment, can reduce insulin sensitivity, highlighting the importance of consistent sleep schedules that correspond with natural circadian rhythms. In a study of adolescents, later bedtimes past 11 PM were associated with impaired insulin sensitivity independent of sleep duration.93 In a separate study, by shifting sleep to daytime hours, insulin sensitivity was reduced by 17% and postprandial glucose increased by 6%, despite maintaining sleep duration.94 Mechanistically, it is believed that impaired sleep contributes to insulin resistance by disrupting the synchronization of circulating hormones, glucose metabolism, and energy utilization. These findings underscore the importance of maintaining a consistent sleep schedule aligned with natural circadian rhythms.
Psychological Health
While sleep deprivation directly impairs insulin function, chronic stress exacerbates these effects through sustained elevations in cortisol and other stress hormones. It is recognized that prolonged psychological stress activates the hypothalamic-pituitary-adrenal axis, leading to increased glucose production and impaired insulin signaling. This stress-induced metabolic dysregulation can further amplify the impact of sleep loss, creating a compounding effect on insulin resistance. Meanwhile, lifestyle modifications that promote stress reduction, such as physical exercise, mindfulness-based practices, breathing exercises, professional counseling, and structured sleep hygiene, can help mitigate these effects.
Practical Strategies to Optimize and Improve Sleep
Below are several strategies that may help to improve sleep quality and sleep duration. All individuals are unique and may respond favorably to some of these strategies, while others will not. The following are intended to be used as tools and techniques to improve sleep quality and duration.
- Establish a Consistent Sleep Schedule: Go to bed and wake up at the same time every day, including weekends. A consistent sleep schedule helps regulate the internal circadian rhythm, improving both sleep quality and overall metabolic health.
- Create an Ideal Sleep Environment: Ensure your bedroom is dark, quiet, and cool. Blackout curtains, white noise machines, earplugs, or eye masks can help minimize disruptions.
- Limit Stimulating Activities Before Bed: In the hour or two leading up to bedtime, avoid stressful or highly engaging activities, such as work-related tasks, social media, or intense discussions. Additionally, it may be reasonable to establish a calming routine with relaxing activities like reading, journaling, deep breathing, or meditation.
- Manage Stress and Anxiety: Chronic stress and unresolved conflicts can significantly impact sleep. Mitigating strategies may include: addressing the root cause of stress when possible; implementing relaxation techniques or structured stress management approaches; seeking professional guidance, such as therapy or counseling.
- Be Mindful of Food, Alcohol, and Stimulants: Avoid large meals, alcohol, caffeine, and nicotine in the hours leading up to bedtime. Late-night food and drink consumption can disrupt sleep by increasing nighttime awakenings and impairing your ability to fall asleep.
- Incorporate Regular Physical Activity: Engaging in daily exercise promotes deeper, more restorative sleep. However, avoid vigorous workouts too close to bedtime, as they may have a stimulating effect.
- Limit Daytime Naps: While short naps (10–20 minutes) can be beneficial, excessive or irregular daytime napping can interfere with nighttime sleep. If you struggle with falling or staying asleep, consider reducing nap frequency and duration.
- Use Relaxation Techniques for Nighttime Awakenings: If you wake up unexpectedly, avoid checking your phone or focusing on the time. Instead, engage in counted breathing – inhale for five seconds, exhale for five seconds – or practice reverse counting to encourage relaxation and prevent the mind from becoming overly active.
- Track Your Sleep Patterns: Maintaining a sleep journal or using a sleep-tracking app can help identify patterns and behaviors that may be disrupting your sleep. Over time, this data can guide personalized adjustments to improve sleep quality.
- Seek Professional Help When Needed: If sleep difficulties persist, consider consulting a sleep medicine specialist or a healthcare professional trained in Cognitive Behavioral Therapy for Insomnia (CBT-I). Additionally, online programs and mobile apps offer accessible and effective tools for sleep optimization.
Supplements and Prescription Medications
In addition to dietary changes and regular physical activity, several classes of prescription medications can improve insulin sensitivity, with many also promoting weight loss. Commonly prescribed options for individuals with type 2 diabetes include Metformin, GLP-1 receptor agonists, SGLT2 inhibitors, and DPP-4 inhibitors (Table 9). Among individuals with insulin resistance but without type 2 diabetes (HbA1c < 6.5%), Metformin is the most frequently used medication.
To compare the effectiveness of Metformin versus lifestyle modifications in preventing diabetes, a study evaluated more than 3,000 adults at risk of developing type 2 diabetes.16 Participants were randomly assigned to one of three groups: a placebo, Metformin, or a lifestyle intervention that included dietary changes and at least 150 minutes of weekly physical activity. Over the study period, the incidence of diabetes was 11.0 cases per 100 person-years in the placebo group, 7.8 cases per 100 person-years in the Metformin group, and 4.8 cases per 100 person-years in the lifestyle intervention group (Table 7). These findings provide compelling evidence that comprehensive lifestyle changes are more effective than medication alone. However, both strategies can be safely used together to further reduce diabetes risk.
Table 7. Incidence of Type 2 Diabetes with Lifestyle Modification and Metformin16
| Placebo | Metformin | Dietary Modification and Physical Fitness | |
| Incidence of Diabetes Per 100 Person-Years | 11 | 7.8 | 4.8 |
| Relative Risk Reduction of Diabetes | – | 31% | 58% |
- Metformin: Improves insulin sensitivity, reducing hepatic glucose production, and enhancing glucose uptake in peripheral tissues. It typically results in a 1-2% reduction in HbA1c and may cause modest weight loss of 1-3 kg, particularly in individuals who are overweight or obese. While not a weight loss drug, it can help improve glucose metabolism and reduce appetite. Common side effects include gastrointestinal issues like nausea, diarrhea, and abdominal discomfort. These side-effects are typically most pronounced when starting or increasing the dose of medication, and often improve with time. Kidney function should be monitored, as Metformin is not safe for use in states of advanced kidney dysfunction.
- GLP-1 receptor agonists (Semaglutide, Tirzepatide): These medications have multiple contributing mechanisms, including the ability to stimulate insulin secretion in response to food, the inhibition of glucagon release, delayed gastric emptying, the promotion of satiety, and decreased caloric intake. These effects contribute to an average HbA1c reduction of 1.5-2.0% and significant weight loss (typically 5-10% of body weight) in individuals with insulin resistance and type 2 diabetes. GLP-1 agonists are particularly effective for individuals seeking to manage weight alongside glucose control. Common side effects include gastrointestinal symptoms like nausea, vomiting, and diarrhea.
- SGLT2 inhibitors (Empagliflozin, Canagliflozin, and Dapagliflozin): By blocking the sodium-glucose cotransporter 2 in the kidneys, this results in increased glucose excretion in the urine. These effects contribute to an average HbA1c reduction of 0.5-1.0% and modest weight loss (typically 2-4 kg). Additionally, SGLT2 inhibitors have benefits in reducing blood pressure and offering cardiovascular and kidney protection, making them useful for individuals with cardiovascular disease or kidney dysfunction. Common side effects include urinary tract infections, genital fungal infections, and dehydration due to the diuretic effect.
- Berberine: Berberine is a natural compound commonly available as an over-the-counter supplement that improves glucose metabolism through several mechanisms. It enhances glucose uptake in peripheral tissues, reduces hepatic glucose production, and increases the expression of GLUT4 in muscle cells, facilitating insulin-independent glucose uptake. Research indicates that berberine can reduce HbA1c by 0.5-1.5%, fasting blood glucose by 9.7 mg/dL, prandial glucose by 16.9 mg/dL, and contribute to modest weight loss (3-5 kg).95 Common side effects include gastrointestinal symptoms such as diarrhea, bloating, and abdominal discomfort. Although generally well tolerated, it is important to consult with a healthcare provider before starting berberine, especially for individuals on other medications, as it may interact with drugs metabolized by the liver.
Table 9. Prescription Medications for the Management of Insulin Resistance
| Medication | Mechanism | Comments | Clinical Indications | Known Side-Effects |
| Metformin (Glucophage) | Increases insulin sensitivity, reduces hepatic glucose production | First-line treatment for type 2 diabetes; effective for lowering blood sugar; often well-tolerated; helps with weight management | Insulin Resistance,Type 2 Diabetes,Polycystic Ovarian Syndrome | Gastrointestinal issues (nausea, diarrhea); rare: lactic acidosis (especially in renal impairment) |
| GLP-1 Receptor Agonists (Ozempic, Wegovy, Trulicity) | Mimics the action of GLP-1 hormone, increases insulin release, decreases glucagon release, slows gastric emptying | Very effective for lowering blood sugar and promoting weight loss; reduces cardiovascular risk | Type 2 Diabetes,Weight Management,cardiovascular risk reductioN | Nausea, vomiting, diarrhea, risk of pancreatitis, potential thyroid cancer risk (rare) |
| SGLT2 Inhibitors (Farxiga, Jardiance, Invokana) | Inhibits sodium-glucose cotransporter 2 in kidneys, increasing glucose excretion in urine | Good for lowering blood sugar; promotes weight loss and reduces blood pressure; beneficial for heart/kidney protection | Type 2 Diabetes,Chronic Kidney Disease, Heart failure | Urinary tract infections, genital infections, dehydration, increased risk of ketoacidosis |
| DPP-4 Inhibitors (Januvia, Onglyza, Tradjenta) | Inhibits the DPP-4 enzyme, increasing GLP-1 levels, which stimulates insulin secretion and inhibits glucagon | Well-tolerated, moderate efficacy in lowering blood sugar; weight-neutral | Type 2 Diabetes | Nasopharyngitis, headache, upper respiratory infections, rare: pancreatitis |
Measurement Outcomes and Tracking Progress
Regularly monitoring biomarkers of health and insulin resistance can provide motivation, reinforce positive lifestyle choices, and ensure safety. Several key biomarkers offer valuable insight into changes in insulin resistance and metabolic health. This includes the LPIR Score, Triglyceride-Glucose Index, and HOMA-IR, which provide a detailed assessment of insulin resistance, while Hemoglobin A1c is particularly useful for individuals with more advanced insulin resistance or type 2 diabetes. Additional indicators, such as body weight, waist circumference, and body composition via whole-body DEXA imaging, help track improvements in overall metabolic health. Blood pressure monitoring is also important, especially for those with hypertension. Finally, ApoB levels should also be regularly checked when making dietary and lifestyle changes.
Importantly, professional oversight by a licensed healthcare provider is essential, particularly for individuals taking insulin or blood pressure medications, as improvements in insulin sensitivity and weight loss often necessitate medication adjustments. Reductions in insulin, diabetes medications, or antihypertensive drugs should always be carefully managed to ensure safety.
Table 10. Comparison of Tests Evaluating Insulin Resistance
| Description | Strengths | Limitations | |
| LPIR Score (Link) | Advanced lipoprotein analysis used to quantify insulin resistance. | Highly sensitive test; identifies insulin resistance among those with and without blood glucose dysregulation; does not rely on fasting glucose or insulin, avoiding variability due to external factors. | Limited awareness and utilization among healthcare professionals; only available at LabCorb. |
| Triglyceride-Glucose Index (Link) | Freely available calculation derived from fasting triglyceride and fasting blood glucose values. | Sensitive test capable of identifying insulin resistance with commonly available test results. | Fasting triglycerides and glucose can fluctuate due to external factors such as acute stress, infection, or recent dietary intake. |
| HOMA-IR(Link) | Freely available calculation derived from fasting insulin and fasting blood glucose. | Sensitive and specific test for identifying insulin resistance. | Requires fasting insulin measurement, which can vary daily due to stress, circadian rhythms, and other non-metabolic factors. |
| Oral Glucose Tolerance Test (OGTT) | Measures glucose and insulin levels at timed intervals after a glucose load (75 g). | Directly evaluates the body’s response to a standardized glucose challenge. | Time-consuming (2-3 hours); fasting required. |
| Hemoglobin A1c (HbA1c) | Measures average blood glucose levels over the past 2-3 months. | Offers long-term insight into blood glucose control; does not require fasting; useful for monitoring advanced stages of diabetes. | Not a direct measure of insulin resistance; influenced by conditions like anemia or altered red blood cell turnover, and factors unrelated to glucose metabolism. |
| Continuous Glucose Monitor (CGM) | Measures real-time glucose levels over days/weeks, capturing fasting, post-meal, and daily glucose variations. | Provides dynamic feedback on glucose trends in relation to food intake, physical activity, and lifestyle. | Does not directly measure insulin resistance; results can vary significantly; primarily designed for diabetes management. |
| Triglyceride:HDL Ratio | A simple ratio of fasting triglycerides to HDL cholesterol. | Easy to calculate; inexpensive; useful for screening for metabolic syndrome and risk of insulin resistance. | An indirect marker of insulin resistance; influenced by factors like dietary habits, physical fitness, and non-metabolic lipid changes. |
Table 11. Advanced Dietary Analytics in the Context of Insulin Resistance
| Total Carbohydrates (grams) | The total amount of carbohydrates in a food, including sugars, starches, and fiber. |
| Net Carbohydrates (grams) | The digestible portion of carbohydrates, calculated as total carbohydrates minus fiber and sugar alcohols (if applicable). |
| Percent Fiber of Carbohydrates | The proportion of total carbohydrates that comes from fiber, calculated as (fiber ÷ total carbohydrates) × 100. |
| Carbohydrate to Protein Ratio | The ratio of carbohydrates to protein in a food or meal, which can influence insulin response and satiety. |
| Glycemic Load | A measure of the blood glucose response to a specific food, calculated as (Glycemic Index × grams of carbohydrate per serving) ÷ 100. It accounts for both the quality (glycemic index) and quantity of carbohydrates consumed. |
| Insulin Index | A measure of how much a food stimulates insulin release, independent of carbohydrate content. It compares the postprandial insulin response of a 240-kcal portion of food to that of a reference food (usually white bread or glucose). Some protein- and fat-containing foods can trigger a higher insulin response than expected based on carbohydrate content alone. |
| Energy Density | The number of calories per gram of food. |






Thanks for the fantastic article. Very interesting and brings up a few interesting questions…
Is an OGTT measuring both glucose and insulin useful and reliable compared to LP-IR, Triglyceride-Glucose Index, and HOMA-IR?
How often should OGTT, LP-IR, Triglyceride-Glucose Index, and HOMA-IR be repeated and monitored in someone who has a family history, is “pre diabetic” and already physically active?
When do you recommend adding Berberine or SGLT2 inhibitor, and is there a specific dose/brand of Berberine or a specific SGLT2 inhibitor you recommend? What do you think about SGLT2s as a general “longevity” drug?
Hi! Thanks for the fantastic article. Very interesting and brings up a few interesting questions…
Is an OGTT measuring both glucose and insulin useful and reliable compared to LP-IR, Triglyceride-Glucose Index, and HOMA-IR?
How often should OGTT, LP-IR, Triglyceride-Glucose Index, and HOMA-IR be repeated and monitored in someone who has a family history, is “pre diabetic” and already physically active?
When do you recommend adding Berberine or SGLT2 inhibitor, and is there a specific dose/brand of Berberine or a specific SGLT2 inhibitor you recommend? What do you think about SGLT2s as a general “longevity” drug?